Semaglutide by Novo Nordisk
Data Focus: Clinical Trial Outcomes and Long-Term Weight Loss Patterns
Introduction:
This page focuses on Semaglutide, a medication developed by Novo Nordisk that has played a significant role in recent obesity research. The purpose of this page is to explore how Semaglutide works, what effects it has on weight loss, and why it matters beyond individual outcomes. By drawing on clinical trial data, research studies, and visualized evidence, this page aims to help readers better understand the medical and social significance of this drug.
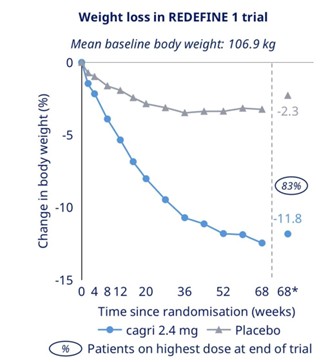
For example, Looking at the chart on the right, weight loss under semaglutide follows a clearly non-linear trajectory rather than a constant rate of decline. The curve shows a steep drop during the early phase of the trial, with the most rapid reduction occurring within the first 16-20 weeks after randomization. After this initial phase, the slope progressively flattens, indicating a marked slowdown in the rate of weight loss. By around week 52 to 68, the curve approaches a plateau, with only minimal additional reduction despite continued treatment. This pattern suggests that semaglutide-induced weight loss reaches a physiological limit over time, rather than continuing indefinitely. While clinical trials demonstrate substantial efficacy, the visualization makes clear that long-term effects are characterized by stabilization rather than ongoing linear loss.
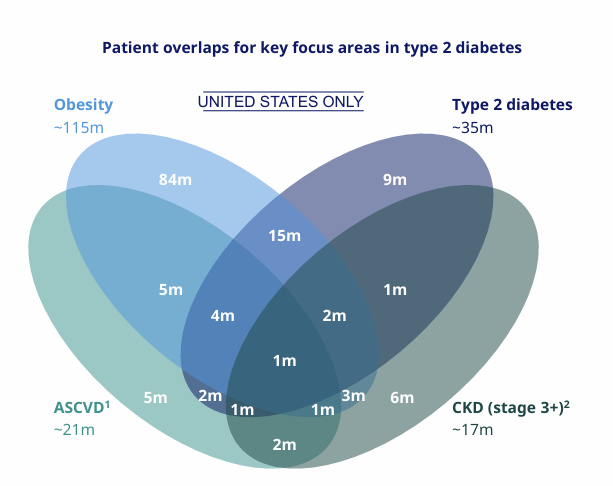
As shown in the diagram, obesity significantly overlaps with type 2 diabetes, cardiovascular disease, and chronic kidney disease. As a result, these drugs are increasingly targeted not only at individuals with obesity, but at patients managing multiple chronic conditions. This expanded target population helps explain why the societal and economic impact of these medications is so large.
Understanding the impact of semaglutide requires combining clinical outcomes, regulatory validation, and manufacturer-provided information. A deeper understanding can be gained by analyzing how these sources complement one another and how users can move between them to answer specific questions about efficacy, safety, and real-world use:
Useful data resources:
-
 Novonordisk.com: the company's official website selling the medicine
Novonordisk.com: the company's official website selling the medicine
- Explains how semaglutide is positioned by the manufacturer, including intended use, dosage structure, and target populations.
- Allows users to see how clinical trial outcomes are summarized and visually communicated to non-specialist audiences.
- Screenshots of product and research pages show how scientific findings are translated into accessible narratives.

 Drugs@FDA: the U.S. Food and Drug Administration's public resource for searching approved drugs, regulatory decisions, and detailed drug approval records.
Drugs@FDA: the U.S. Food and Drug Administration's public resource for searching approved drugs, regulatory decisions, and detailed drug approval records.
- Provides regulatory approval records, official labeling, and clinical review summaries.
- Enables users to verify manufacturer claims and identify approved indications, dosage limits, and known side effects.
- Screenshot examples demonstrate how users can trace regulatory decisions and safety evaluations.
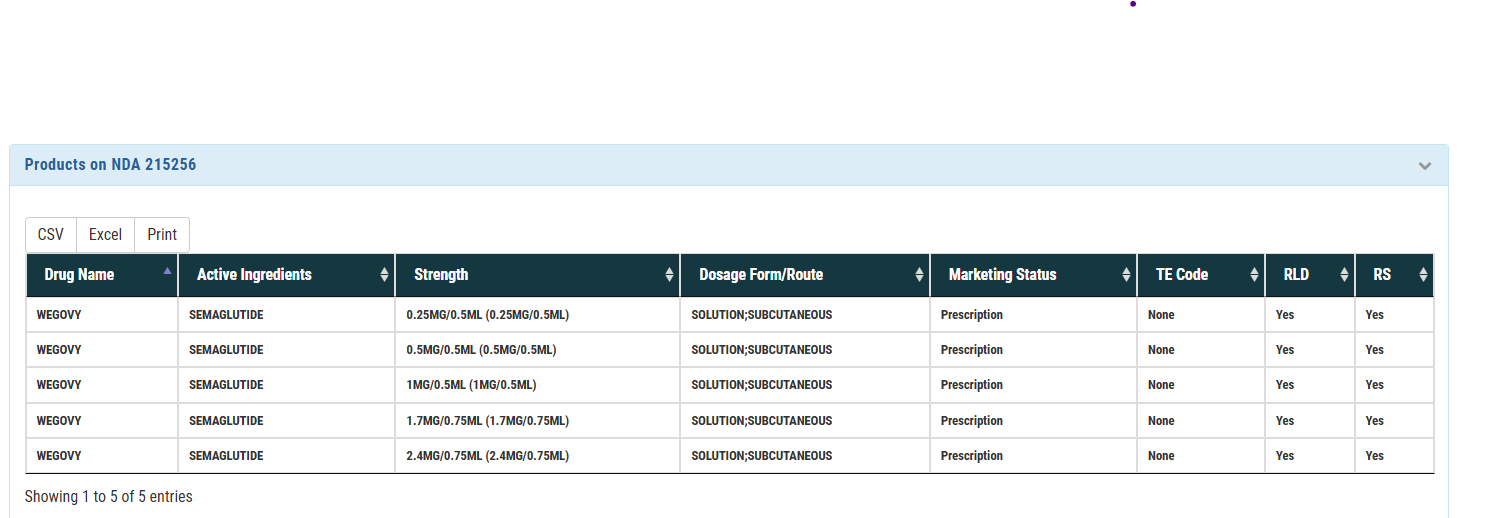
 The New England Journal of Medicine:a leading peer-reviewed medical journal publishing high-impact clinical research, reviews, and commentary on medicine and public health.
The New England Journal of Medicine:a leading peer-reviewed medical journal publishing high-impact clinical research, reviews, and commentary on medicine and public health.
- Offers peer-reviewed clinical trial publications with detailed study design and outcome data.
- Allows users to analyze long-term effectiveness patterns, such as changes in weight-loss rate over time.
- Screenshots from trial figures and results illustrate how raw data and statistical findings are presented.
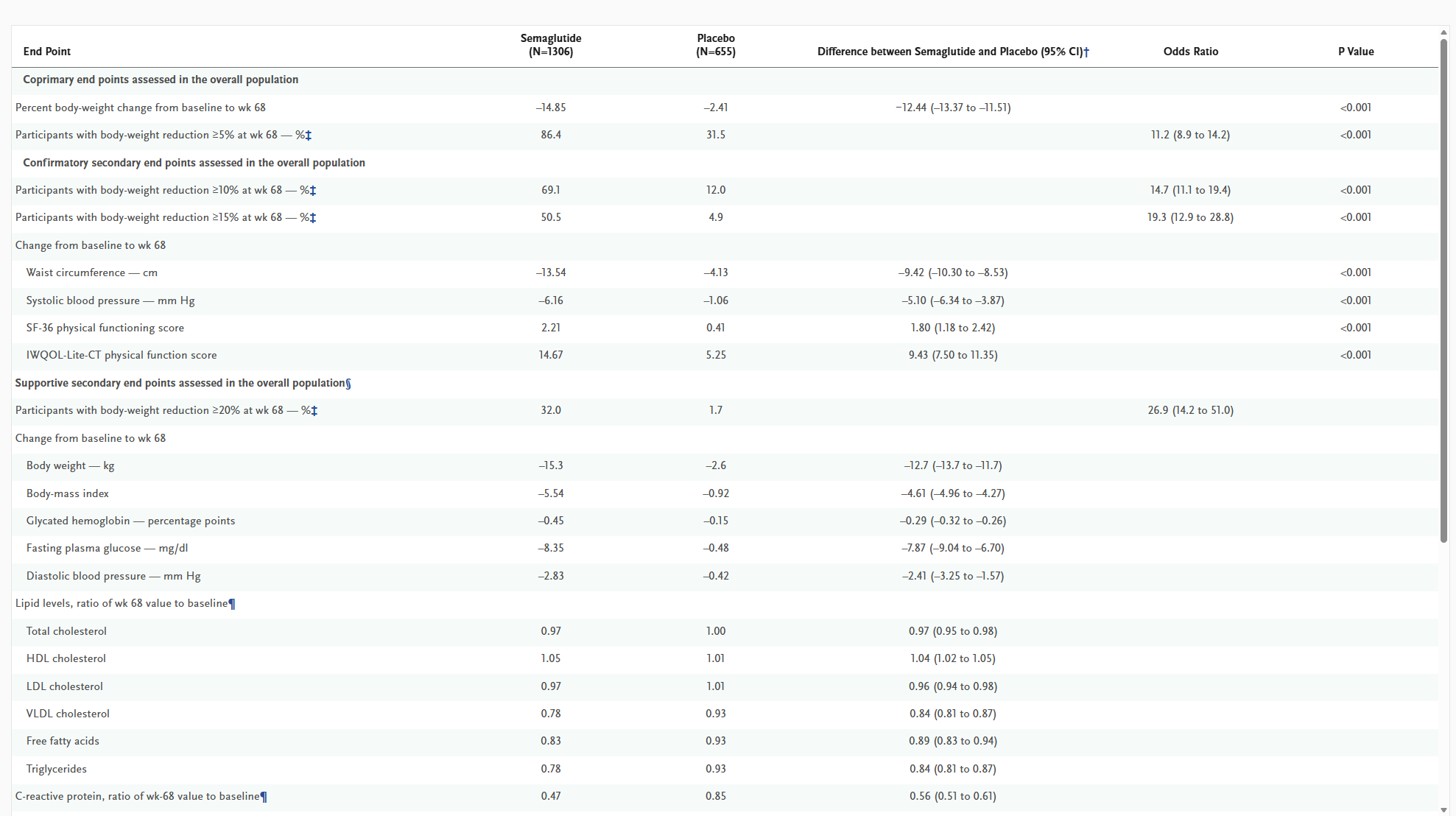
Sample Analysis--Understanding the Long-Term Effectiveness of Semaglutide:
Whether Semaglutide Leads to sustained weight loss over time?
- Examine long-term clinical trial data
- The first step is to consult peer-reviewed clinical trial results published in The New England Journal of Medicine. Trial figures allow users to observe how body weight changes over time under semaglutide treatment, including whether weight loss continues linearly or stabilizes.
- By the New England Journal of Medicine articles
- Verify approved use and safety considerations
- After identifying patterns in clinical trial data, users can turn to Drugs@FDA to verify how these findings translate into approved medical use. The FDA database provides official labeling, dosage limits, and safety evaluations that contextualize the trial results within regulatory decisions.
- By Drugs@FDA
- Compare with manufacturer communication
- Finally, users can review how Novo Nordisk presents semaglutide on its official website. Comparing manufacturer summaries with clinical and regulatory data helps users understand how complex trial results are translated into accessible narratives for non-specialist audiences.
- At Novo Nordisk Product and Research Page